We do not diagnose disease or recommend a treatment protocol or dietary supplement for the treatment of disease. You should share this information with your physician who can determine what nutrition and disease treatment regimen is best for you. Ask your physician any questions you have concerning your medical condition.
You can search this site or the web for topics of interest that I may have written (use Dr Simone and topic).
“We provide truthful information without emotion or influence from the medical establishment, pharmaceutical industry, national organizations, special interest groups or government agencies.” Charles B Simone, M.MS., M.D.
85% OF LIVES COULD HAVE BEEN SAVED IF EARLY INEXPENSIVE TREATMENT WITH HYDROXYCHLOROQUINE OR IVERMECTIN WERE USED
https://tinyurl.com/4637kyfy Charles B. Simone, M.MS., M.D. Written Dec 18, 2023
Three major studies that were given as testimony in the U.S. Senate and the Texas State Senate showed that 85% of lives could have been saved if early inexpensive treatment with hydroxychloroquine or ivermectin were used.



IVERMECTIN IS OVER-THE-COUNTER IN 3 STATES AND MORE STATES TO FOLLOW
As of July 10, 2025, 4 states passed laws allowing for OTC distribution of ivermectin. Tennessee was the first state to make the drug available in 2022 with SB 2188/HB 2746, which authorized “ivermectin [as] suitable for human use.”
Arkansas passed SB 189 and Act 369 on March 25, 2025, and soon after, on April 14, Idaho passed SB 1211. Both states and their respective acts indicate that “ivermectin [is] suitable for human use [and] may be sold or purchased as an over-the-counter medication without a prescription or consultation with a health care professional.”
Louisiana is the latest state to join the movement for legislation of OTC ivermectin. On June 20, 2025, SB 19 and Act 464 went into effect.
Other states are following suit—passing legislation to expand public access to ivermectin. These include3:
-
-
-
Texas: HB 3219 would reclassify ivermectin as OTC.
-
New Hampshire: SB 119 would pair ivermectin status with an unrelated Medicaid provision and “…[direct] pharmacists to dispense brand-name drugs to Medicaid beneficiaries when the brand-name drug is on the Department of Health and Human Services preferred drug list.”
-
North Carolina: HB 618 would “issue a statewide standing order to allow pharmacists to dispense ivermectin without prescription” and “[provide] immunity from civil or criminal liability” for any pharmacist who dispenses ivermectin.
-
Pennsylvania: SB 601 would amend the state’s Pharmacy Act to include OTC ivermectin.
-
Georgia: SB 366 would “provide for ivermectin as an over-the-counter medication in this state” and “remove ivermectin from the definition of, exceptions to, and exemptions from dangerous drugs.”
-
Alabama: HB 278 would “permit a physician, a physician assistant, or a certified registered nurse practitioner to give a pharmacist a standing order that has been approved by the State Board of Pharmacy to dispense ivermectin to individuals without a prescription.” This law includes similar requirements to the Louisiana bill. Additionally, it includes disciplinary protections for pharmacists and restricts them from endorsing the use of ivermectin to an individual or the public.
-
West Virginia: SB 614 would authorize pharmacists to sell ivermectin without a prescription or consultation, as well as protect them from liability from damages or licensing board discipline.
-
Missouri: SB 744 would permit pharmacists to dispense both ivermectin and hydroxychloroquine OTC.
-
South Carolina: HB 3916 would allow OTC sales of ivermectin and hydroxychloroquine without a prescription or consultation.
-
-
In 2005 the CDC published a paper entitled – “Chloroquine is a potent inhibitor of SARS coronavirus infection and spread.” Virol J. 2005 Aug 22;2:69.
February 2020 – Some members of the Covid-19 Task Force want to use hydroxychloroquine (HCQ), a safe and effective FDA-approved drug, to control the rapidly spreading pandemic. The drug has been shown to prevent hospitalization if taken within the first five days of COVID-19 symptoms. HCQ costs about 60 cents per 200 mg tablet – treatment is one tablet twice a day for five days – $6.00 The FDA said HCQ is safer than Tylenol.

https://www.cnn.com/videos/politics/2020/04/22/hydroxychloroquine-trump-covid-19-study-mh-orig.cnn


June 29, 2020 – The Henry Ford COVID-19 Task Force in Detroit studied 2,541 patients (International Journal of Infectious Diseases) entitled, Treatment with Hydroxychloroquine, Azithromycin, and Combination in Patients Hospitalized with COVID-19. https://www.ijidonline.com/action/showPdf?pii=S1201-9712%2820%2930534-8 (journal pre-proof).
Conclusion: “Among hospitalized patients, use of hydroxychloroquine alone and in combination with azithromycin was associated with a significant reduction in-hospital mortality compared to not receiving hydroxychloroquine.”
January 28, 2021 – FACEBOOK and AMERICAN JOURNAL OF MEDICINE admit their position on hydroxychloroquine was wrong.
Early use of hydroxychloroquine had a 79% lower mortality rate in countries that allowed the use of the safe drug.
A new study in the American Journal of Medicine in January showed a much lower mortality rate for patients with COVID-19 being treated with hydroxychloroquine while still at home. Also, the Facebook Oversight Board said it overturned its previous case on the effectiveness of HCQ.
HYDROXYCHLOROQUINE SAVES LIVES https://bit.ly/38t2ZKl
IVERMECTIN SAVES LIVES https://bit.ly/3aCNAtl reviews many studies
Meta-analyses based on 18 randomized controlled treatment trials of ivermectin in COVID-19 have found large, statistically significant reductions in mortality, time to clinical recovery, and time to viral clearance. In summary, based on the totality of the trials and epidemiologic evidence presented in this review along with the preliminary findings of the Unitaid/WHO meta-analysis of treatment RCTs and the guideline recommendation from the international BIRD conference, ivermectin should be globally and systematically deployed in the prevention and treatment of COVID-19.
WHO SUPPRESSED IVERMECTIN’s USE
1) MEDIA –
New York Times – “Senate hearing promoted unproven drugs and dubious claims about ivermectin.”
AP – “No evidence ivermectin is a miracle drug against COVID-19”
2) BIG TECH –
YouTube, owned by Google – banned ivermectin information from Dr Risinger and Dr. Martenson.
Twitter – blacklisted European Medical Journal for published study on ivermectin.
Facebook – removed posts of Front Line Covid-19 Critical Care
3) NIH – formed a panel of doctors who decided what treatments can be given to patients. Ivermectin was not on the list and, in fact, the panel recommended use Remdesivir as highly recommended by Fauci – “diminishes the time to recovery.” Midway through the study, the endpoint was changed from mortality to recovery because Remdesivir does not save lives. The World Health Organization does not recommend it at all. Gilead owns Remdesivir. Seven members of the panel are paid by Gilead, including the three co-chairs of the panel who choose the panel members.
2015 – During a National Academies of Science Workshop Peter Daszak said this, “until an infectious disease crisis is very real, present, and at an emergency threshold, it is often largely ignored. To sustain the funding base beyond the crisis, a key driver is the media and the economics follow the hype. Investors will respond if they see profit at the end of the process.”
Nov 9, 2015 – COVID-19 Birth Announcement in Nature Medicine. The COVID-19 Bioweapon was created in plain sight. This new intentionally made chimeric virus “shows a gain in pathogenesis“ (page 1512, middle of third paragraph).
Jan 9, 2017 – a few days before leaving office OBAMA RESUMED FUNDING for GAIN-OF-FUNCTION
Jan 10, 2017 – the next day Fauci said “THERE WILL BE A SURPRISE OUTBREAK”
https://rumble.com/v1dgdhp-fauci-there-will-be-a-surprise-outbreak-january-10-2017.html
Jan 20, 2017 – Trump is inaugurated
2018 – The NIH infected 12 bats with a SARS-like coronavirus. The virus was shipped from Wuhan to an NIH lab in Montana (DailyMail https://dailymail.co.uk/health/article-12693441/fauci-nih-infected-bats-camp-david-coronavirus.html
September 18, 2019 – W.H.O. said by 2020 there would be an accidental or voluntary release of a respiratory pathogen and a global simulation so that we would accept a universal global vaccination.
October 18, 2019 –  – the global simulation – hosted by Gates Foundation in partnership with the World Economic Forum, and the Bloomberg School of Public Health Johns Hopkins. Co-hosted by Avril Haines, former director of the CIA and today the Director of National Intelligence.
– the global simulation – hosted by Gates Foundation in partnership with the World Economic Forum, and the Bloomberg School of Public Health Johns Hopkins. Co-hosted by Avril Haines, former director of the CIA and today the Director of National Intelligence.

October 29, 2019 — 11 days later – Panel at the Michael Milken Institute Future of Medicine, discusses how to force and implement a new vaccine.
Listen especially to Rick Bright of BARDA (Biomedical Advanced Research and Development Authority of HHS) who said “a novel avian flu virus from China could allow them to push through a new universal flu vaccine much faster.”
https://youtu.be/sl_a-x09PQg
https://rumble.com/v18s8tv-october-29-2019-panel-discusses-how-to-force-and-implement-vaccine.html
November 17, 2019 – China records first known case of COVID-19 but US Senate Report states first death from COVID in China occurred in September 2019.
December 12, 2019 – NIH gives Ralph Baric the mRNA prototype to make the vaccine – this prototype was made weeks/months before Wuhan scientists had symptoms. It is synthetic modified messenger RNA (mRNA) that is more stable, immunogenic, and potent.
January 21, 2020 — CDC – first U.S. COVID Case
WE HAD A NATIONAL PANDEMIC RESPONSE PLAN FOR SUCH AN EMERGENCY that was initially tested in 2000, revised in 2005, and updated in 2017 https://www.hsdl.org/?abstract&did=438701
WHY WAS IT NOT FOLLOWED?
HHS Secretary Alex Azar, formerly of Big Pharma Eli Lilly
Jan 27, 2020 – Determination that a Public Health Emergency Exists based on confirmed cases of 2019 Novel Coronavirus – however, there was no test to confirm the cases, nor was the virus even named yet. W.H.O. named virus on February 11, 2020
https://www.phe.gov/emergency/news/healthactions/phe/Pages/2019-nCoV.aspx
Under section 319 of the Public Health Service Act, the Secretary of Health and Human Services (HHS) may declare the existence of a public health emergency (PHE) such as a substantial outbreak of an infectious disease. Once a declaration is in place, the Secretary can take various actions, including making grants and contracts and waiving or modifying certain requirements of Medicare, Medicaid, the Children’s Health Insurance Program, and the Health Insurance Portability and Accountability Act.
HHS issued four section 564 declarations, allowing the FDA to issue EUAs for in vitro diagnostics, personal respiratory protective devices, other medical devices, and drugs and biologic products (including vaccines). A medical product can be made available under an EUA under four conditions: if the agent referred to in the declaration can cause a serious or life-threatening disease or condition; if there is no adequate, approved, and available alternative; if the product may be effective; and if the known and potential benefits of use of the product outweigh the known and potential risks. https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/mcm-emergency-use-authorities
February 4, 2020 – Azar declares 2019 Novel Coronavirus is a threat to national security
President Clinton defined national security: provide for the common defense, promote the general welfare, and secure the blessings of liberty to ourselves and our posterity. A NATIONAL SECURITY STRATEGY FOR A GLOBAL AGE https://history.defense.gov/Portals/70/Documents/nss/nss2000.pdf
February 2020 – Some members of the Covid-19 Task Force want to use hydroxychloroquine (HCQ), a safe and effective FDA-approved drug, to control the rapidly spreading pandemic. The drug has been shown to prevent hospitalization if taken within the first five days of COVID-19 symptoms. HCQ costs about 60 cents per 200 mg tablet – treatment is one tablet twice a day for five days – $6.00
The FDA said HCQ is safer than Tylenol.

https://www.cnn.com/videos/politics/2020/04/22/hydroxychloroquine-trump-covid-19-study-mh-orig.cnn

March 19, 2020 – President Trump advocates hydroxychloroquine.
March 20, 2020 – Fauci says “No. Only anecdotal evidence.”
Louisiana Attorney General Jeff Landry deposed Fauci for 7 hours in November 2022. Fauci said that Hydroxychloroquine was “dangerous” and had “toxic” side effects…. Fauci claimed HCQ was ineffective in treating COVID, but couldn’t cite a single study to support his claim. Fauci also rejected the list of 371 studies on HCQ and its effectiveness in treating the disease when he was presented with the list. https://brownstone.org/articles/faucis-7-hour-deposition-what-we-know-so-far/
March 23, 2020 – President Trump advocates hydroxychloroquine.
March 23 and 24, 2020 – Rick Bright PhD of HHS, Janet Woodcock MD of FDA defied their orders and ignored and intentionally sabotaged this Plan by preventing the distribution of hydroxychloroquine to the public as they were ordered, but instead forced hydroxychloroquine to be used only in hospital settings for advanced illness, thus allowing the virus to spread rapidly.
They told the media that hydroxychloroquine was not safe and not effective. They and Anthony Fauci MD, and a biased media destroyed the comprehensive National Pandemic Response Plan that would have promoted early outpatient antiviral drug treatment. The deaths of hundreds of thousands of Americans and the destruction of the U.S. economy could have been prevented.
Rick Bright PhD of HHS, Janet Woodcock MD of FDA (Acting FDA Commissioner 1-20-21 to 2-17-22) manipulated the system to prevent the distribution of hydroxychloroquine to the public under an Investigational New Drug (IND) authorization as they were ordered, but instead forced hydroxychloroquine to be used only in hospital settings for advanced illness under an Emergency Use Authorization (EUA), thus allowing the SARS-CoV-2 virus to spread rapidly. They told the media that hydroxychloroquine was not safe and not effective, got its EUA revoked, and got an EUA for an unsafe and ineffective tax-payer funded Remdesivir.
March 26, 2020 – Fauci, Lane, and Redfield (CDC) wrote: “This suggests that the overall clinical consequences of Covid-19 may ultimately be more akin to those of a severe seasonal influenza (which has a case fatality rate of approximately 0.1%) or a pandemic influenza (similar to those in 1957 and 1968) rather than a disease similar to SARS or MERS, which have had case fatality rates of 9 to 10% and 36%, respectively.

Covid-19 — Navigating the Uncharted https://www.nejm.org/doi/full/10.1056/nejme2002387
March 27, 2020 – President Trump advocates hydroxychloroquine.
March 27, 2020 – Azar declared the Emergency Use Authorization of drugs and biological products during the COVID-19 pandemic
March 29, 2020 – President Trump advocates hydroxychloroquine.
April 3, 2020 – President Trump advocates hydroxychloroquine.
April 4, 2020 – President Trump advocates hydroxychloroquine.
April 4, 2020 – Fauci appears to be unaware of the National Pandemic Plan and refused to consider the use of HCQ for Covid-19 treatment. He dismisses the accumulating HCQ efficacy reports from China, South Korea and France as simply ‘anecdotal’ and diverts millions to Remdesivir.
April 5, 2020 – President Trump advocates hydroxychloroquine.
April 10, 2020 – President Trump advocates hydroxychloroquine.
April 13, 2020 – President Trump advocates hydroxychloroquine.
April 22, 2020 – Rick Bright is fired for insubordination.
April 24, 2020 – FDA warns there are potentially deadly side effects from HCQ and it is not safe and not effective for COVID-19.
May 18, 2020 – President Trump says he is taking hydroxychloroquine.
In 2005 the CDC published a paper entitled – “Chloroquine is a potent inhibitor of SARS coronavirus infection and spread.” Virol J. 2005 Aug 22;2:69.
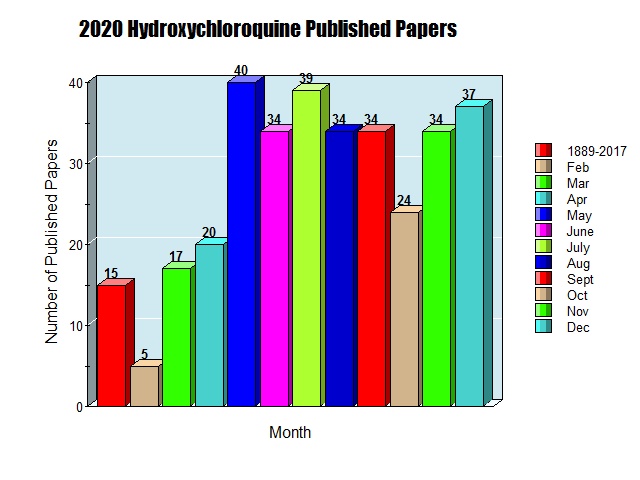
Of the 372 total published papers showing that chloroquine / hydroxychloroquine is effective against SARS coronavirus, 22 were published during February and March 2020 and scores and scores more were published during 2020.

https://c19hcq.org/ – This Site provides a complete list of the published peer-reviewed papers.
FTX, a Bahamas-based cryptocurrency exchange and a partner with the World Economic Forum, filed bankruptcy November 2022, CEO Sam Bankman-Fried. FTX also funded and disseminated information via TogetherTrials.com that hydroxychloroquine and ivermectin were not effective. The TogetherTrials were given Trial of the Year Award from the Society for Clinical Trials in San Diego (May 16, 2022).


May 22, 2020 – A Lancet article was RETRACTED but stated: Hydroxychloroquine or chloroquine decreased in-hospital survival and an increased frequency of ventricular arrhythmias when used for treatment of COVID-19.
Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. https://doi.org/10.1016/S0140-6736(20)31180-6

This intentional sabotage of the validated National Pandemic Response Plan paved the way for the “vaccine.”
Despite the long list of known possible adverse events, the FDA grants EUA to both Pfizer and Moderna
December 11, 2020 – FDA granted an EUA for the Pfizer–BioNTech COVID‑19 vaccine.
December 18, 2020 – FDA granted an EUA to Moderna (mRNA-1273)
The CDC had to change its definition of a vaccine on November 4, 2021 to justify its needs.
Original definition: “A product that stimulates a person’s immune system to produce immunity to a specific disease, protecting the person from that disease.”
NOW, “A preparation that is used to stimulate the body’s immune response against diseases.”
Next came an invalid Emergency Use Authorization (EUA) for the “vaccine” – an EUA should only be granted if there was a “novel” or new feature to the virus that presents as a new threat that can be isolated, identified, evidenced, and supported by clear data.
1) the virus was not novel because there were 73 patents on parts of this virus and scores of medical papers, and
2) there were effective treatments
Also, Obama removed SARS from the Biological Select Agents and Toxins List (BSAT) because SARS – Coronavirus – no longer posed a threat to humans.
The following is an important summary of the harm of the “vaccines”
https://x.com/DrSimone/status/1722338317818024223?s=20
THE COVID “VACCINE” WAS NOT EFFECTIVE AND WAS NOT SAFE. AND IT DID NOT SAVE MILLIONS OF LIVES. https://x.com/DrSimone/status/1722290948846109158?s=20…
This information has been taken with permission from
COVID-19 BIOWEAPON² PART 1 Executive Summary https://bit.ly/3Fuiwdu
Written: March 2020
BIOWEAPON 1 – virus
BIOWEAPON 2 – shots
COVID-19 BIOWEAPON² PART 2 https://bit.ly/3eI11bK
COVID-19 BIOWEAPON² PREREQUISITE FOR U.S. INVASION – LONG TERM PLANNING
https://tinyurl.com/37fbrevd
COUNTERMEASURE “VACCINE” – NOT EFFECTIVE, NOT SAFE, NOT MADE AT “WARP SPEED”
https://tinyurl.com/5n7tajc5
CHILDREN SHOULD NOT GET THE “VACCINE”
“ABOVE ALL DO NO HARM”
Written November 21, 2021 https://bit.ly/3nB4CyK
PROTECT YOURSELF FROM THE SPIKE: COVID-19 INFECTION and/or VACCINE – OUR NEXT HEALTH CRISIS
Written August 23, 2021 https://bit.ly/3kkrejY