We do not diagnose disease or recommend a dietary supplement for the treatment of disease. You should share this information with your physician who can determine what nutrition, disease and injury treatment regimen is best for you. You can search this site or the web for topics of interest that I may have written (use Dr Simone and topic).
“We provide truthful information without emotion or influence from the medical establishment, pharmaceutical industry, national organizations, special interest groups or government agencies.” Charles B Simone, M.MS., M.D.
CLINICAL TRIAL SPONSORS KEEP RESULTS SECRET AND DON’T PAY BILLIONS IN FINES
https://www.simonesuperenergy.com/wp-content/uploads/2020/02/CLINICAL-TRIAL-SPONSORS-KEEP-RESULTS-SECRET-AND-DONT-PAY-BILLIONS-IN-FINES-1.wav
Lawrenceville, NJ (Dr Charles Simone) – The Food and Drug Administration and the National Institutes of Health wrongly allow clinical trial sponsors to keep their results secret and escape from paying more than $12 billion in fines since the law was enacted in 2007 requiring disclosure of results. This is more than $13,000 per day in fines for not reporting results. Any group that conducts clinical trials are required to record their results within one year of trial completion on the NIH’s website ClinicalTrials.gov. Because of its common format, this site, that has 215 million monthly page views, allows for easy comparison of results. Physicians and patients rely on trial results to see whether new treatments are safe and effective.
The FDA and NIH can enforce the law at any time – but they have not. The FDA can impose the daily fine that amounts to more than $12 billion owed to the American taxpayer. And the NIH can stop funding those who ignore the law – but they have not.
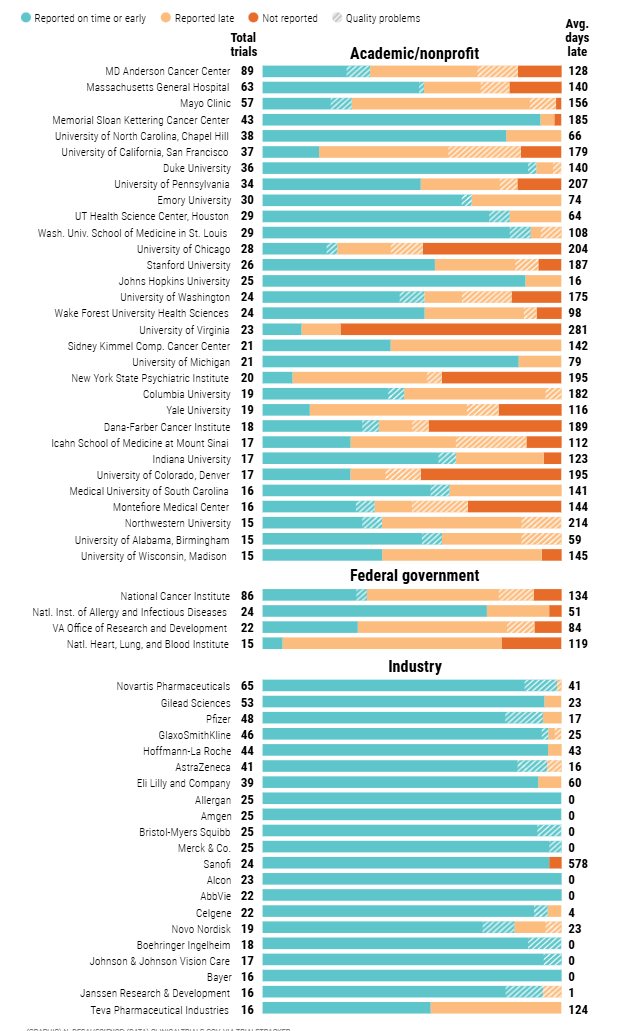
More than 4700 clinical trials were reviewed in the journal Science: 45% reported results on time, 32% did not report at all, and 23% reported late. They include universities, NIH itself, and more than 20 Big Pharma companies. https://www.science.org/content/article/fda-and-nih-let-clinical-trial-sponsors-keep-results-secret-and-break-law. In April 2026, the FDA sent more than 2,200 warning letters involving over 3,000 clinical trials that appeared noncompliant.
Failure to report negative or inconclusive studies distorts medical evidence, influences prescribing patterns, and undermines informed patient care. Some top offenders:
Major Organizations Cited in Reporting Analyses
1). Academic / Nonprofit Institutions
-
-
-
-
MD Anderson Cancer Center
-
Massachusetts General Hospital
-
Mayo Clinic
-
Memorial Sloan Kettering Cancer Center
-
University of California San Francisco
-
Duke University
-
University of Pennsylvania
-
-
-
2). Federal Agencies
-
-
-
-
National Cancer Institute
-
National Institute of Allergy and Infectious Diseases
-
National Heart Lung and Blood Institute
-
VA Office of Research and Development
-
-
-
3). Big Pharma
-
-
-
-
Pfizer
-
Novartis
-
Gilead Sciences
-
GlaxoSmithKline (GSK)
-
AstraZeneca
-
Eli Lilly and Company
-
Amgen
-
Bristol Myers Squibb
-
Merck and Company
-
Hoffman LaRoche
-
-
-
Science analyzed ClinicalTrials.gov records of all clinical trials with results legally required to be reported between 18 January 2018 and 25 September 2019. The chart, covering trial sponsors with 15 or more results due in that window or reported early, shows that some results deposited were not posted due to quality lapses.